Breathing New Life Into Lung Cancer Research
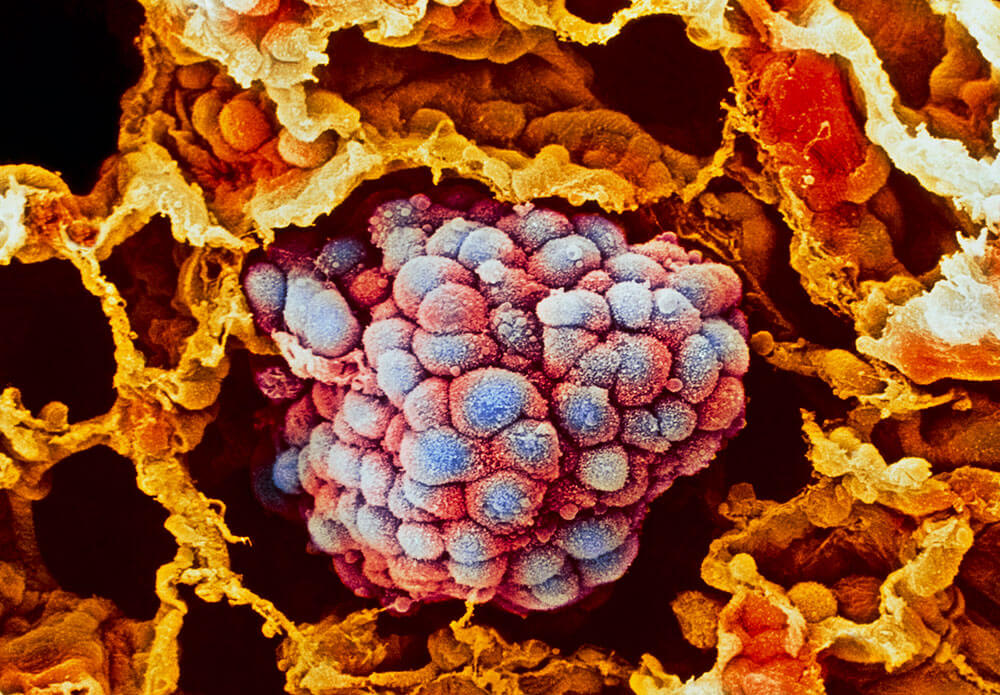
The story of cancer was long seen as a single narrative: a cell acquires genetic mutations, starts to divide uncontrollably and then spreads.
But in recent years, this tale has become more complex. Scientists are increasingly aware that a tumor’s microenvironment — its surrounding healthy cells, including fibroblasts that form connective tissues, as well as blood vessels and immune cells — are more than just innocent bystanders, they actively contribute to the developing tumor and can play a role in encouraging its growth or keeping it at bay.
To better understand the role of the tumor microenvironment, scientists have been working in recent years to develop cell culturing techniques that can grow cells in 3-D using scaffolding matrices. The traditional 2-D models that grow cancer cells in a flat, plastic dish do not capture this biological complexity. While several 3-D models have been widely used in the field, scientists, until recently, were unable to create a 3-D model for a hard-to-treat subtype of lung cancer known as squamous cell carcinoma.
