Matching Drugs to Diseases: Advances in Targeting JAKs Leading to New Autoimmune Therapies
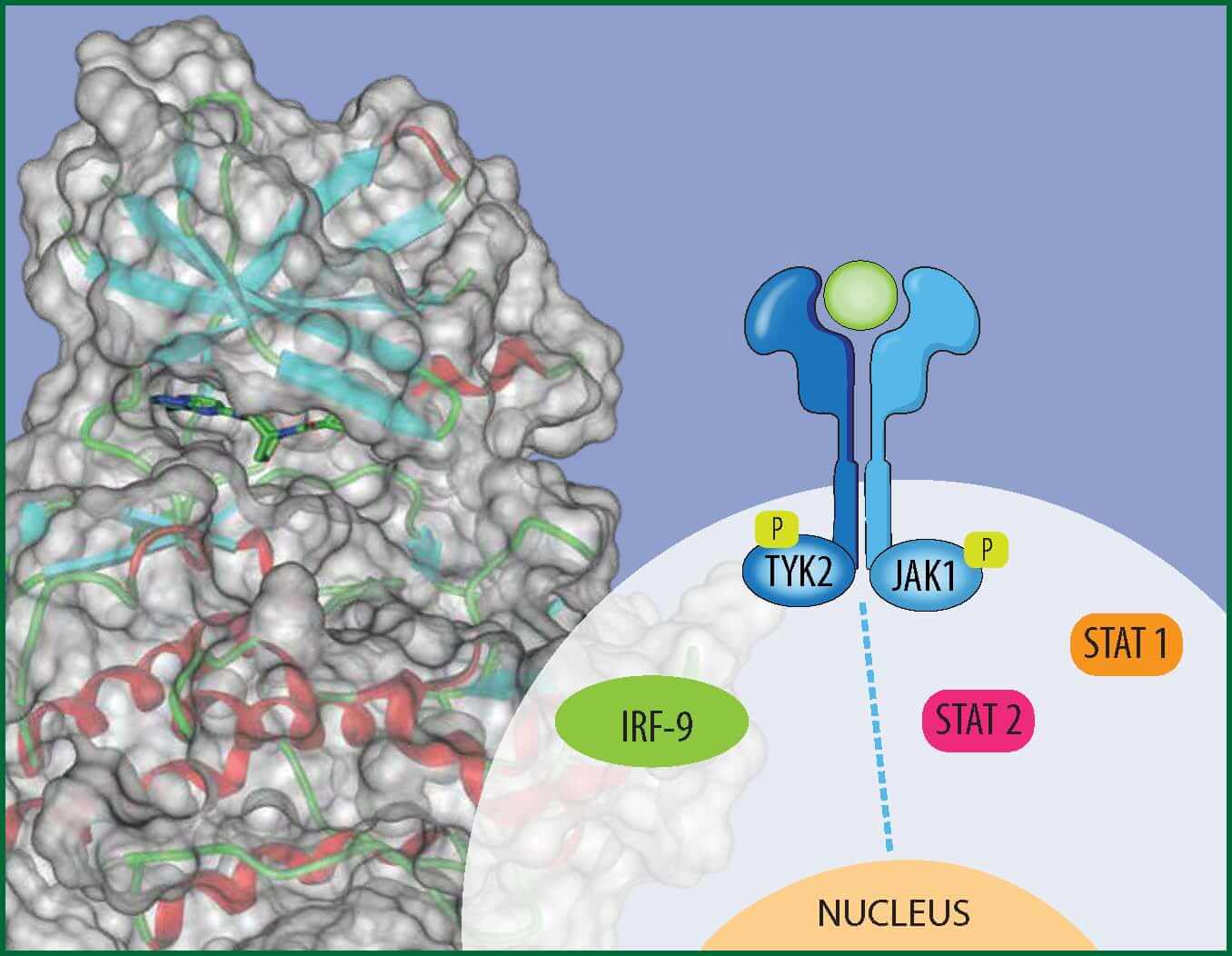
Janus kinases (JAKs) named after Janus, the Roman two-faced god of duality, are enzymes with two “faces,” or domains, that play a key role in signal transduction of cytokines, the molecular messengers that trigger the inflammatory and immune responses. Scientific discoveries related to JAKs in recent years have led to novel treatments for autoimmune diseases and myeloproliferative disorders, a group of conditions where the bone marrow produces too many blood cells.
In the early 2010s, the first class of oral JAK inhibitors emerged for treating autoimmune conditions like rheumatoid arthritis, psoriasis and inflammatory bowel disease, and also for myeloproliferative disorders. These medicines work by blocking several members of the JAK family — JAK1 and JAK3 and to a lesser extent, JAK2 — which in turn reduces the overactive cytokine response that leads to inflammation, causing autoimmune diseases.
As scientists have increased their understanding of the JAK signaling pathways, they hope to develop new medicines that can target members of the enzyme family to different degrees. A team of Pfizer scientists have made breakthroughs in developing a portfolio of potential new medicines that selectively inhibit JAK1, JAK3 and TYK2, and dual inhibition of TYK2 and JAK1. The discovery story behind the TYK2 and JAK1 dual inhibitor was recently published in the Journal of Medicinal Chemistry.
“Autoimmune diseases are driven by a group of pro-inflammatory cytokines and other signaling molecules that lead to activation of subsets of inflammatory cells,” says Andy Fensome, an Associate Research Fellow at Pfizer’s Kendall Square research site in Cambridge, Mass. “By focusing on molecules that block different JAKs to differing degrees, we hope to increase the benefit to patients by targeting more specific pathways important in different diseases, but avoiding blockade of signals that we think are not important in those diseases.”
Cytokine Selectivity
The key to the recent breakthroughs is an increased understanding of the overactive cytokines that drive the inflammatory cycle in different autoimmune diseases. Coupling that understanding with the knowledge of which JAK enzymes are critical for these cytokine pathways allowed scientists to design more targeted small molecule drugs.“It’s all about hitting multiple pathways in diseases that are driven by multiple cytokines,” says Fensome. “And understanding the similarities between disease pathologies can allow us extrapolate.”
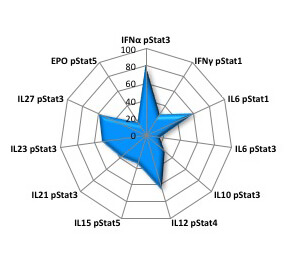
Since different autoimmune diseases are driven by distinct cytokines, understanding the cytokine inhibition profile of a molecule can determine how well it might treat a disease. Shown here is the cytokine inhibition profile for the JAK1/TYK2 inhibitor, showing the extent to which the small molecule interferes with the expression of specific cytokines. Courtesy Andy Fensome.
So why JAK1 and TYK2? Dual inhibition of both of these kinases puts into motion a blockade of multiple pro-inflammatory cytokines. And these cytokines come from families known to be important in specific diseases because of their role in specific inflammatory cascades. For example, studies with biologics have demonstrated that IL-23 signaling is a key component of the TH17/IL-17 proinflammatory cascade, which is important in both psoriasis and Crohn’s disease. In addition to the TYK2-driven IL-23 and IL-12 signaling pathways, researchers have also identified the JAK1-mediated common gamma-chain, interferon-gamma, and IL6 families as key mediators of these and other autoimmune diseases.
JAK enzymes are believed to play an important role for over 50 cytokines and growth factors, many of which drive immune-mediated conditions. Further, additional kinase enzymes beyond JAKs are believed to be important in cytokine signaling, providing further opportunities for targeted inhibitors in the treatment of autoimmune disease. By understanding the complex relationships between JAKs and other kinases, cytokines and immune cell responses, scientists can potentially develop more targeted therapies across a broad range of autoimmune conditions.
